Merck Covid Pill : E3edk3fjwhk4am
Pharmaceutical company Merck on Friday said it will seek an emergency authorization for their oral antiviral. 1 2021 that its experimental COVID-19 pill reduced hospitalizations and deaths by half in people recently infected with the coronavirus and that it would soon ask health officials in the US.
An antiviral pill developed by US.

Merck Covid pill. Merck said it would ask US regulators to authorise the first antiviral pill to treat Covid-19 after a late-stage clinical trial showed the drug cut the risk of hospitalisation or death in half. Drugmaker Merck Co could half the chances of dying or being hospitalized for those most at risk of contracting severe COVID-19 with experts hailing it as a. Merck and partner Ridgeback Biotherapeutics plan to seek US.
Merck Co. Said Friday that its experimental Covid-19 pill reduced hospitalizations and deaths by half in people recently infected with the coronavirus and that it would soon ask health officials. News Merck says antiviral COVID-19 pill halves death and hospitalizations.
Merck Co. Merck found its antviral pill molnupiravir can reduce pandemic hospitalizations and deaths by half in a test thats yet. Said Friday that its experimental COVID-19 pill reduced hospitalizations and deaths by half in people recently infected with the coronavirus and that it would soon ask health officials.
An antiviral pill developed by US drug maker Merck could halve the chances of dying or being hospitalised from COVID-19 but experts warn vaccines are still necessary. Government with its pill for Covid-19 should the experimental drug receive regulatory clearance. Entered a 12 billion agreement to supply the US.
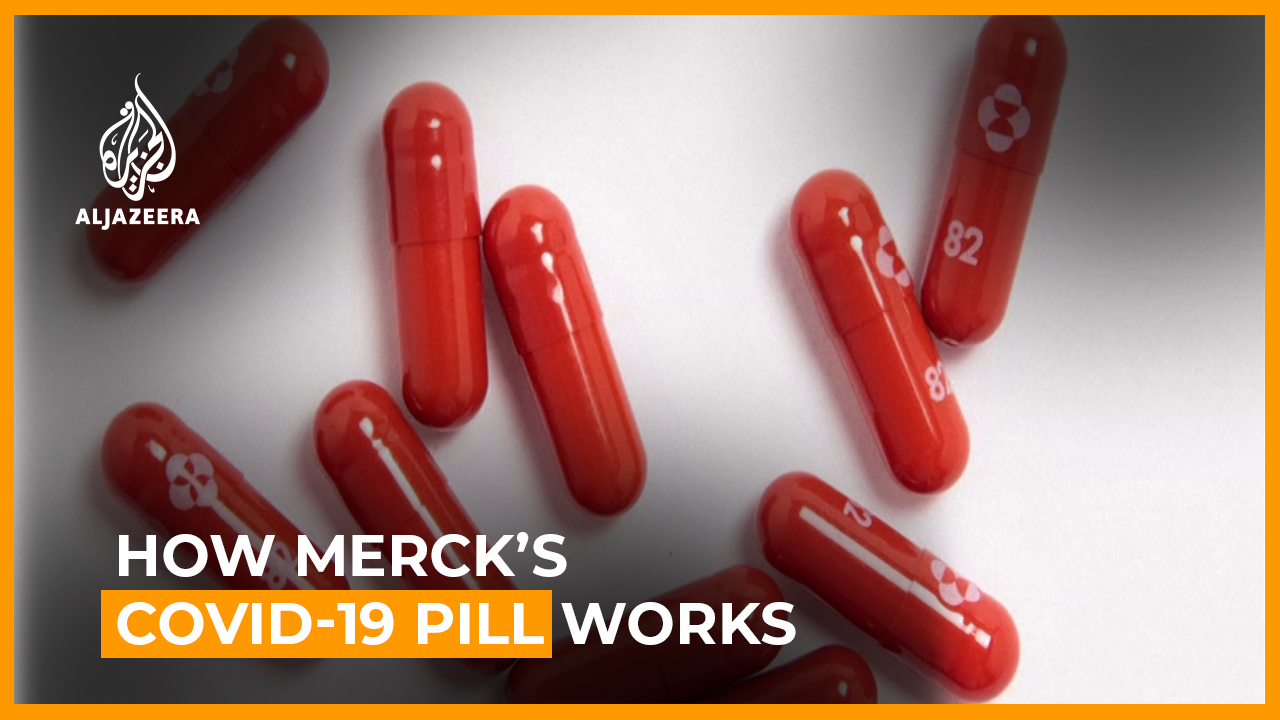
Mercks antiviral pill reduces hospitalization of Covid patients a possible game-changer for treatment. Drugmaker Merck says its experimental COVID-19 pill reduced hospitalizations and deaths by half in people recently infected with the. Both Pfizer and Merck are working on a so-called COVID pill that could help treat people with the virus who arent at risk of developing severe illness.
Said Friday that its experimental COVID-19 pill reduced hospitalizations and deaths by half in people recently infected with the. Merck on Friday announced that its new pill to treat Covid-19 reduced the risk of hospitalization and death by about 50 percent. Merck is conducting a late-stage trial to see if its antiviral pill can prevent COVID-19 infection in addition to the study that showed it can significantly cut hospitalization and death in those.
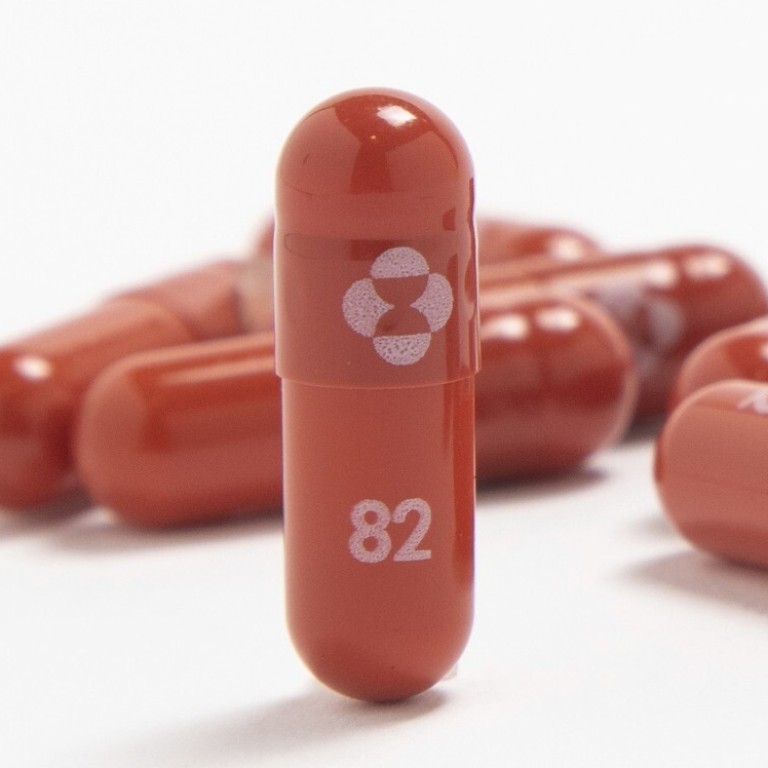
Merck Cos Covid-19 antiviral pill molnupiravir reduced the risk of hospitalization or death by 50 in an interim analysis of a late-stage trial findings that could give doctors another. This undated image provided by Merck Co. Mercks COVID Pill Cuts Risk of Death Hospitalization by 50 Interim Study Shows.
A pill to treat Covid-19. Shows their new antiviral medication. Pharmaceutical company Merck Co.
Merck says COVID-19 pill cuts death risk in half will seek emergency authorization. Washington Merck Co. Merck and its partner Ridgeback Biotherapeutics said early results showed patients who received the drug called molnupiravir within five days of COVID-19 symptoms had about half the rate of hospitalization and death as patients who received a dummy pill.
At the interim analysis molnupiravir reduced the risk of hospitalization or death by approximately 50 Merck. Merck says COVID-19 pill cuts risk of death hospitalization. Merck Co.
And around the world to authorize its use. Merck says experimental pill cuts worst effects of COVID-19 Health Canada working to review Mercks experimental COVID-19 pill treatment These. Were talking about a return to maybe normal life.
Merck agreed to supply 17 million. Emergency use authorization for their oral drug for COVID molnupiravir as soon as possible.

Merck Mrk Molnupiravir Pill Could Change The Fight Against Covid Bloomberg

From Pfizer To Shionogi Drugmakers Race For Coronavirus Pills Nikkei Asia





:no_upscale()/cdn.vox-cdn.com/uploads/chorus_image/image/69938987/AP21274373443876_copy.0.jpg)